How many of you all couldn’t recognize your friends after the quarantine period? Out of the many, you might have held your tummy in! Lack of proper exercise is observed in 8 out of 10 teenagers making them “Fat”, which is an unpleasant word for most. But the facts on fats are more diverse and are an essential selection through the evolution of life.
Fats
Lack of knowledge on fats had made a huge impact on the well-being of society. People tend to not buy products with high-fat content. But isn’t that good? Actually, it’s not the amount of fat that causes fatness, and other diseases. It’s the type of fats.
A fat molecule consists of glycerol and 3 fatty acid chains. Glycerol is more like the backbone of fats. The fatty acid chains with their long carbon chains can either contain single bonds or double bonds between the carbon atoms. They are saturated fatty acids and unsaturated fatty acids respectively.
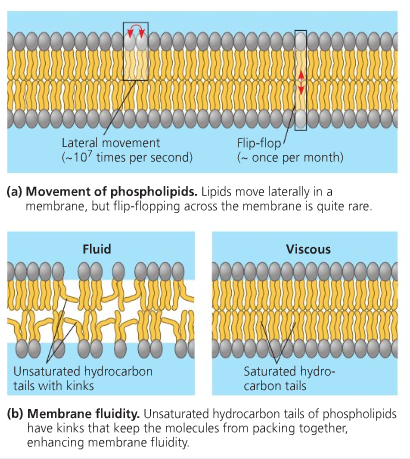
Saturated fatty acids consist of straight carbon chains and they pack tightly together being fully extended. Their compactness allows them to be solids at room temperature e.g. butter. When Carbon atoms have double bonds, they are unsaturated. A carbon-carbon double bond can exist in two forms as cis configuration and trans configuration.
Considering unsaturated fatty acids with a cis double bond, a bend is formed in the hydrocarbon chain, as a result making them less compact compared to fully saturated fatty acids. Since they are already poorly ordered, it takes less energy to melt them. Hence their melting point is lower than that of saturated fatty acids. There can be mono and polyunsaturated fatty acids. Unsaturated fatty acids in the cell membrane facilitate transport and cellular signaling, which exists in an optimum Unsaturated/Saturated fatty acid ratio.
Omega-3 fatty acids and Omega-6 fatty acids are considered essential fatty acids because they cannot be synthesized by humans.
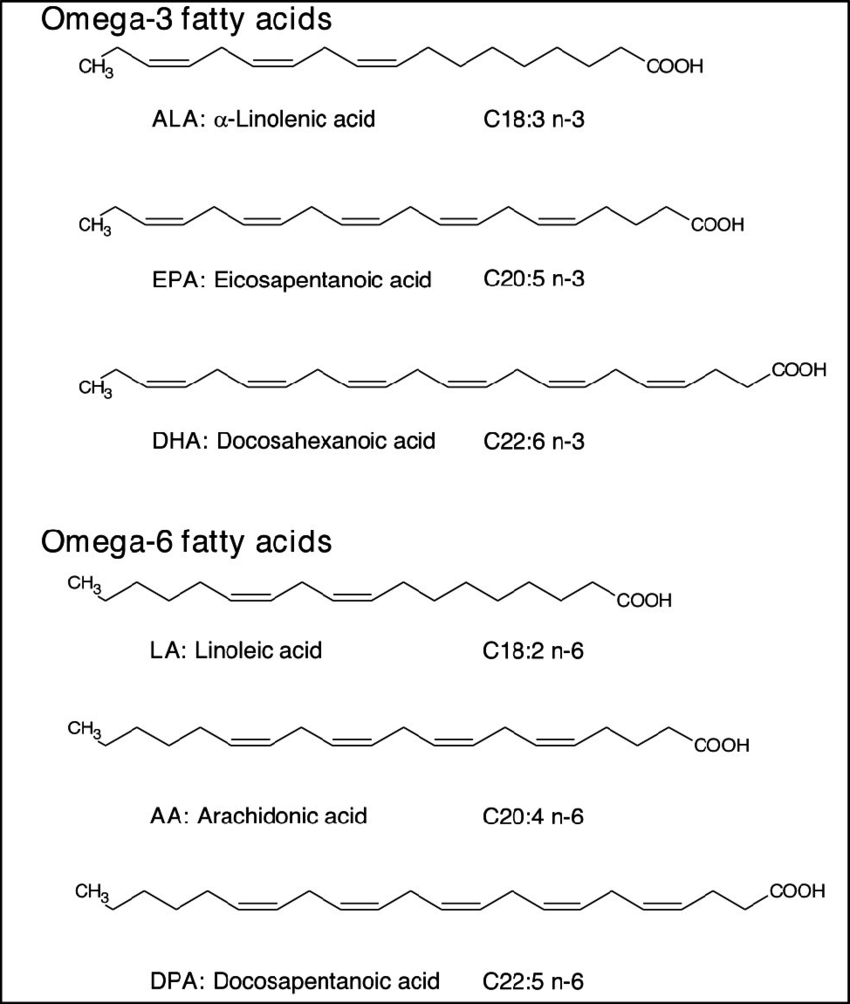
The Good Fats
Studies reveal that saturated fats increase LDL by inhibiting LDL receptor activity in the Liver. LDL receptors act like a harbor where LDL can dock into. The inhibition of their activity leads to an increased concentration of LDL in the bloodstream. Unsaturated fats, on the other hand, enhances the LDL receptor activity and decreases the LDL concentration in the bloodstream. Both saturated and unsaturated fatty acids increase HDL in the bloodstream.
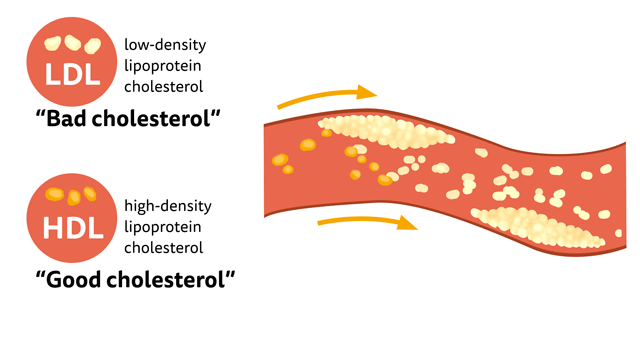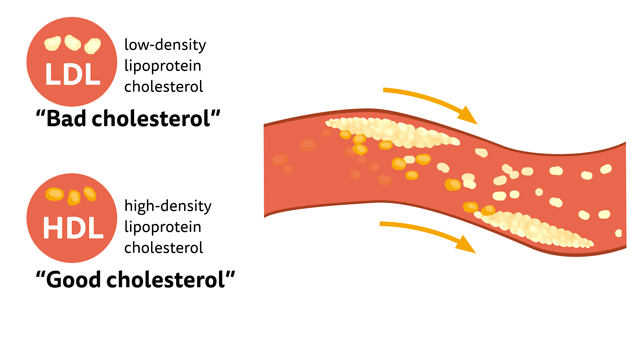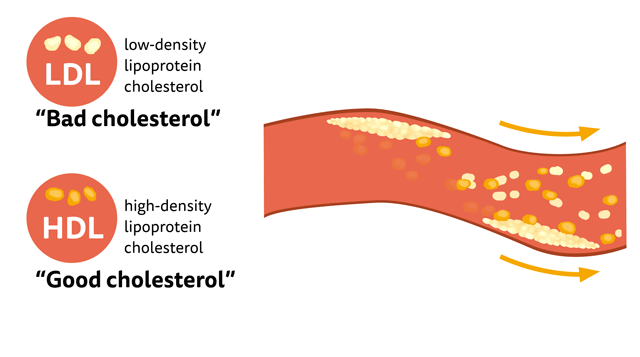
What is “good cholesterol”?
There is a common misconception that Cholesterol is bad for our body, In fact, a distinct amount of cholesterol is needed to maintain cell membranes, produce hormones, Vitamin D, and bile acids that assist in digesting facts. LDL is considered to be “bad cholesterol” at high concentrations than the desired level, since they accumulate in the walls of arteries forming plaque, particularly in the coronary artery which increases the risk of heart attacks. HDL, on the other hand, is like a vacuum cleaner that removes excess cholesterol, plaque and carries it to the liver to be removed. Hence the term “good cholesterol”.

Formation of trans fatty acids
Simply, unsaturated fatty acids can be converted to saturated fatty acids by hydrogenation. It resembles the addition of hydrogen to alkene resulting in an alkane in the presence of a catalyst.
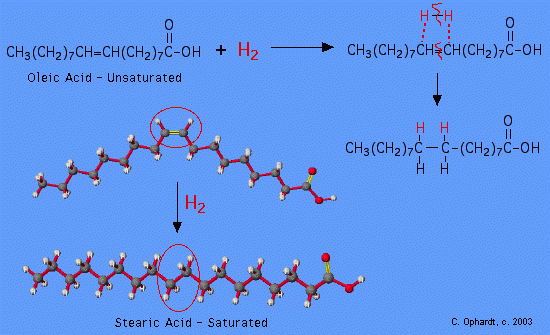
Vegetable oils are mostly polyunsaturated fats with several Carbon-Carbon double bonds. They are less stable and have high solubility. Food manufacturers convert these liquids to solids or semi-solids to harden them and increase shelf time e.g. margarine. This is achieved by a process called partial hydrogenation where the polyunsaturated fats are now partially saturated, increasing the melting point. During this process, something else happens.
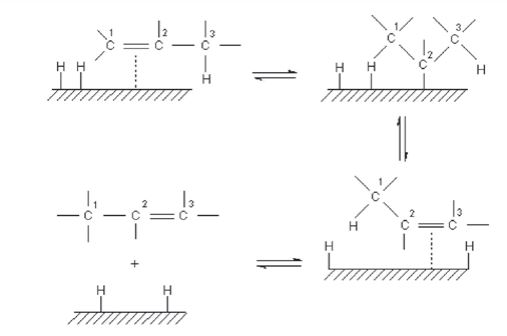
Trans fats are produced as a result of a side reaction with the catalyst of the hydrogenation process. As discussed previously trans and cis are isomers of unsaturated fat, having the same molecular formula but bonded differently.
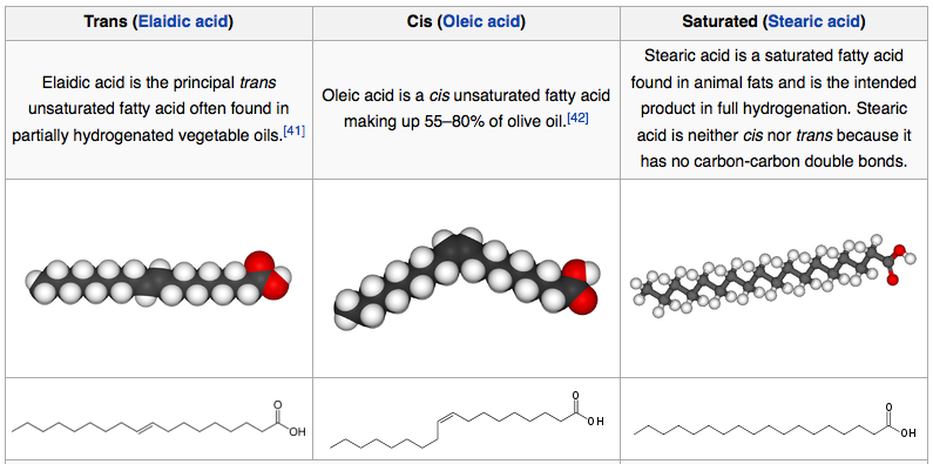
Trans fats rarely occur in nature and therefore the body does not have the necessary enzymes to recognize them and break them down. Trans fats raise LDL and lower HDL branding themselves as unhealthy fats. Further Trans fats cause blood platelets aggregation, increasing cell membrane rigidity, and decreasing liver detoxification capacity thus increasing the chance of heart diseases and cancers.
From the Grocery store
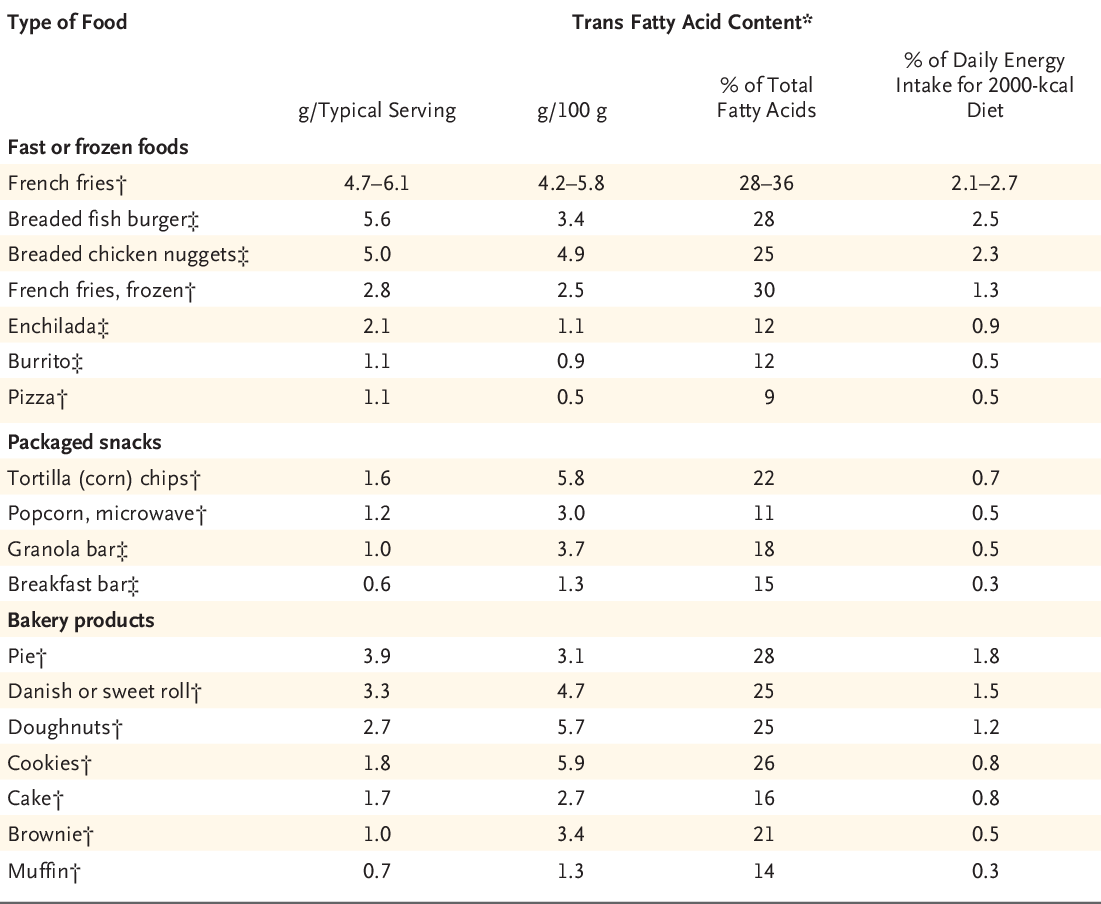
When considering olive oil and pancake mix, where olive oil has nearly 10 times the fat percentage than that of pancake mix, yet olive oil is good for our health. This is because nearly half of the composition of fat in pancake mix is either trans fat or saturated fats, but totally unsaturated fats in olive oil. The occurrence of trans fat in food products can be identified by the term “partially hydrogenated” in the ingredient list.
In summary, the fact that matters is not how much fat we eat, but the type of fat we eat, and which fats are good for our health are determined by their shape.
Image Credits:
- Feature image: https://bit.ly/3syZxrc
- Header animation: https://bit.ly/35A2o9p
- Fig.1: https://bit.ly/3bHETir
- Fig.2: https://bit.ly/3oJvIBT
- Fig.3: https://bit.ly/3oJBe7A
- Fig.4: https://bit.ly/3nKpy33
- Fig.5: https://bit.ly/2XFcJwo
- Fig.6: https://bit.ly/2XClPtA
- Fig.7: Snip from https://bit.ly/38HYvkx
- Fig.8: https://bit.ly/2LPtfae
- Fig.9: https://bit.ly/35FMQkj
References:
- https://chem.libretexts.org/Bookshelves/Biological_Chemistry/Supplemental_Modules_(Biological_Chemistry)/Lipids/Fatty_Acids/Hydrogenation_of_Unsaturated_Fats_and_Trans_Fat
- https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2943062/#:~:text=Studies%20in%20animals%20have%20shown,containing%20lipoprotein%20production%20%5B6%5D.
- https://www.researchgate.net/publication/244750076_Fatty_acid_contents_of_Brazilian_soybean_oils_with_emphasis_on_trans_fatty_acids
- https://www.youtube.com/watch?v=QhUrc4BnPgg